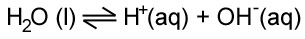IMPORTANCE OF WATER
|
WATER STRUCTURAL MOLECULE
HYDROGEN BOND IN WATER MOLECULE
|
IONIZATION OF WATER
|
THERMAL AND SOLVENT PROPERTIES IN WATER
1. MODERATION OF TEMPERATURE BY WATER
|
- Evaporation water loss functions:
- Help to cool body temperature.
- Stabilize temperature in lakes and ponds.
- Help to keep tissues in leaves from becoming too warm in sunlight.
2. EXPANSION UPON COOLING
|
- Advantages of water expansion upon cooling:
- During winter, the water freeze into ice and float on the surface, allowing the life to exist below the frozen surface.
- Ice provide solid habitat for animals such as polar bear and seals.
3. VERSATILITY AS SOLVENT
- Water is a universal solvent.
- Solvent is the dissolving agent. Substance that is dissolved is solute.
- When solute is dissolve in solvent, the mixture is called solution.
- For example, a salt water is a solution, having water as the solvent and salt (NaCl) as solute.
- Dissolving of salt in water occurs when hydrogen regions is attracted to chloride ions of salts.
- Water molecules will surround the individual chloride ion, forming a sphere called hydration shell.
- A compound does not need to be ionic to dissolved in water.
- Non-ionic polar molecules such as sugar are also water-soluble.
- Large molecules such as proteins can dissolved in water if they have ionic and polar regions on their surface.
- Biological fluids, such as blood, the sap of plant, liquid within the cells are having the properties of water dissolving polar compounds.